The Michaelis–Menten equation describes how the rate of an enzyme-catalysed reaction increases with substrate concentration and eventually plateaus at a maximum value () due to enzyme saturation.
Consider an enzyme binding reversibly to a substrate
to form the Michaelis complex
, which then produces product(s)
and regenerates the enzyme:
where ,
and
are rate constants.
The rate of formation and breakdown of is given by:
Let’s assume:
-
- The initial rate method is used to measure the reaction.
- The initial substrate concentration
is much greater than the initial enzyme concentration
, so that the free substrate concentration
is approximately equal to
.
- Initial product formation is negligible and the step
is irreversible.
- Total enzyme concentration is conserved:
.
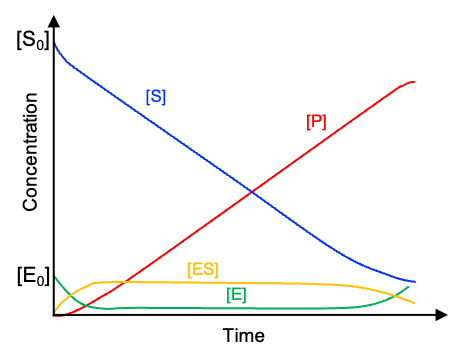
Therefore, the enzyme–substrate complex builds up rapidly during an initial transient phase and then reaches a constant concentration (see diagram above). During this period, . This is known as the steady-state approximation, with
where is the Michaelis constant.
Substituting into eq80 gives:
which rearranges to:
Since ,
Because product formation is relatively slow initially, becomes the rate determining step, with the initial reaction velocity
of the overall reaction being:
Substituting eq81 into eq82 yields the Michaelis-Menten equation:
where is a constant for a fixed
.
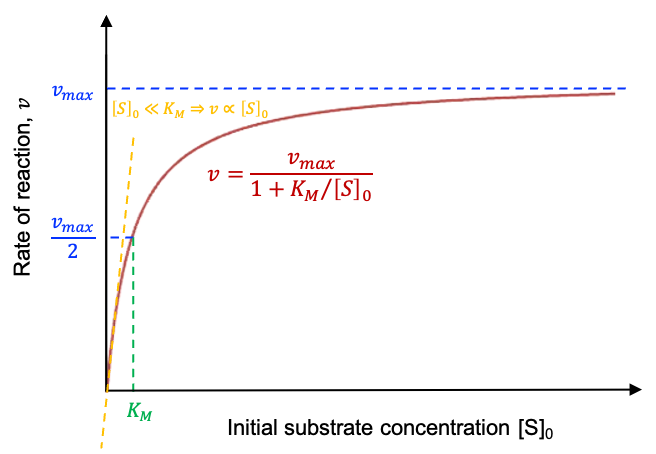
When , we have
(see above diagram). In other words, the rate of the enzyme-catalysed reaction reaches a maximum when the substrate concentration is in excess. Here, the reaction is zero-order with respect to the substrate concentration.
Rearranging eq83 to shows that at low initial substrate concentration (
), the reaction approximates first-order kinetics for a fixed
:
.
When , we have
.
At a specific temperature and pH, is characteristic for a given enzyme-substrate pair. It can be interpreted as a measure of the affinity of the enzyme for its substrate because it can be rewritten as:
Here, is the equilibrium constant for
. A smaller
(and hence a smaller
) corresponds to greater the affinity of the enzyme for its substrate, since the enzyme–substrate complex is less likely to dissociate back into free enzyme and substrate. Additionally, because
is equal to the substrate concentration at which the reaction rate is half-maximal, a smaller
means the enzyme can achieve this rate at lower substrate concentrations and therefore operates more efficiently.

Question
What is the definition of catalytic efficiency?
Answer
Catalytic efficiency is defined as , which is equivalent to
because
. The term
corresponds to the maximum number of reactions each enzyme molecule can catalyse per unit time. Furthermore, a smaller
indicates a higher affinity of the enzyme for its substrate. Therefore,
captures both the catalytic speed and substrate-binding efficiency of the enzyme, with a larger value indicating higher catalytic efficiency.
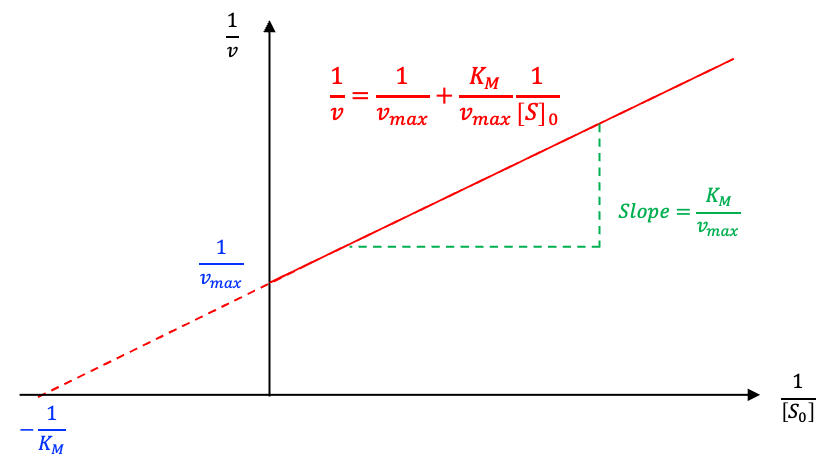
To determine experimentally,
is rearranged into a Lineweaver-Burk form:
A plot of against
for different initial substrate concentrations results in a straight line with intercept
and slope
, from which
can be determined (see diagram below).