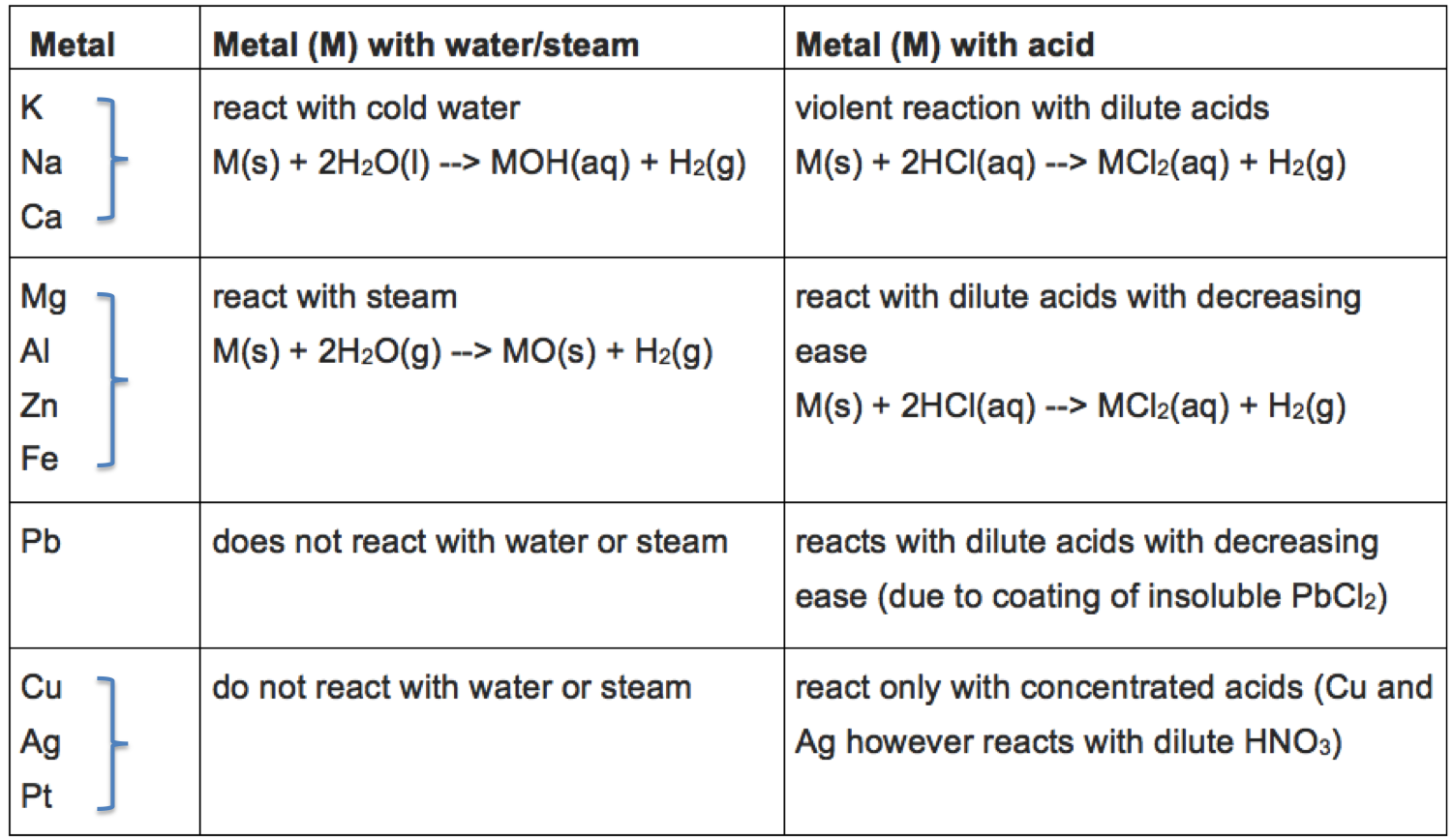
Some of the reactions of metals with water and acid are summarised in the table below.
May the mole be with you
Some of the reactions of metals with water and acid are summarised in the table below.

The reactivity of metals broadly refers to the relative extent of change that metals undergo with respect to certain reactions. Some of these reactions are:
In general, the greater the extent to which a metal changes from its elemental form to a compound, the more reactive it is. For example, sodium is more reactive than copper, as elemental sodium reacts violently with cold water, while elemental copper does not react with water and steam. The reactivities of metals for reactions 1 to 5 are summarised in the following table, which is known as the metal reactivity series:
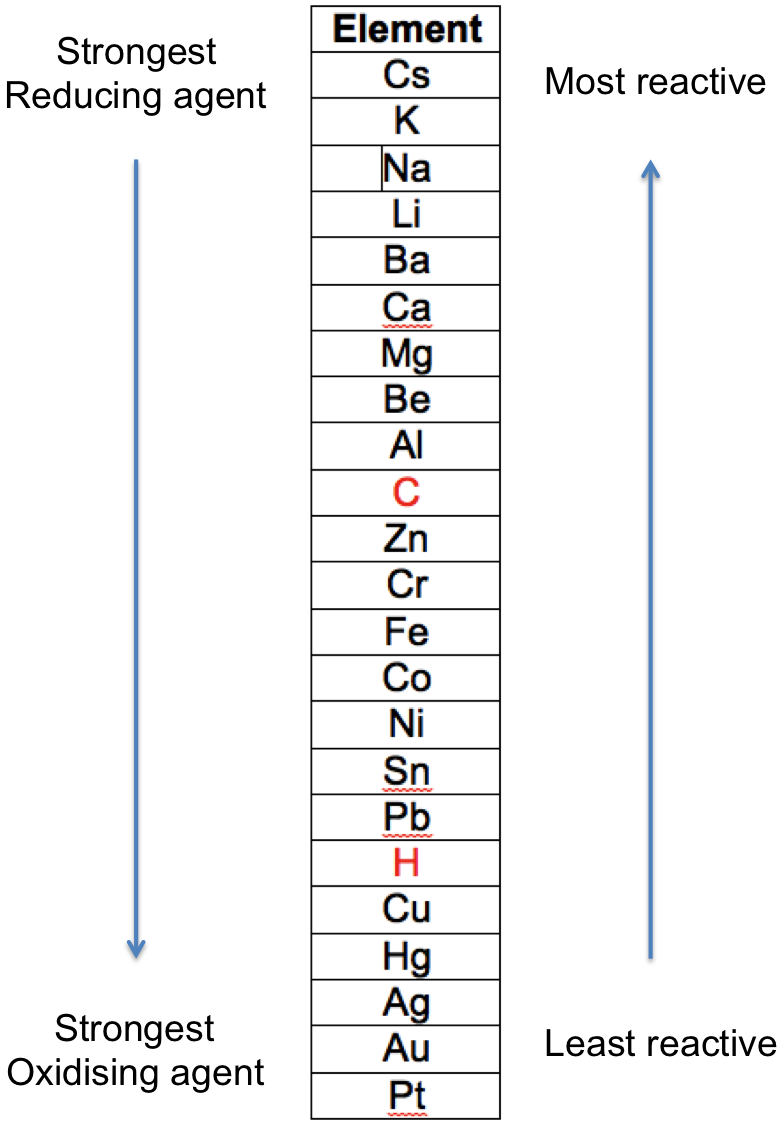
The two non-metals, C and H are included for reference.
What are the effects of pH on redox reactions?
pH affects the strength of an oxidising agent. For example, the oxidising strength of potassium manganate (VII), as measured by its standard electrode potential, is as follows:
Acidic medium
MnO4– + 8H++ 5e– → Mn2+ + 4H2O Eored = +1.51 V
Neutral/slightly basic medium
MnO4– + 2H2O + 3e– → MnO2 + 4OH– Eored = +1.23 V
Strongly basic medium
MnO4– + e– → MnO42- Eored = +0.56 V
Hence, potassium manganate (VII) is most strongly oxidising in an acidic medium and least oxidising in a strongly basic medium.
The behaviour of MnO4– in different pH conditions is best explained using the the reaction MnO4– + 2H2O + 3e– → MnO2 + 4OH– as a reference. If the pH of this reaction is lowered, excess H+ allows manganese dioxide to be further reduced to the more stable +2 oxidation state ion:
MnO2 + 4H++ 2e– → Mn2+ + 2H2O
Combining the half reactions MnO4– + 2H2O + 3e– → MnO2 + 4OH– and MnO2 + 4H++ 2e– → Mn2+ + 2H2O gives the overall reduction reaction of MnO4– to Mn2+ in acidic medium.
On the other hand, if the pH of the same reaction MnO4– + 2H2O + 3e– → MnO2 + 4OH– is increased, excess OH– will shift the equilibrium towards the left. As the solution becomes more strongly basic, the manganate (VII) ion is instead reduced to the manganate (VI) ion. This implies that the manganate (VI) ion is stable only in very basic conditions. It disproportionates in acidic, neutral and slightly basic conditions as follows:-
3MnO42- + 4H+ → 2MnO4– + MnO2 + 2H2O
Incidentally,
| MnO4– | purple |
| MnO42- | green |
| MnO2 | brown solid |
| Mn2+ | pale pink |
How do we construct and balance redox equations?
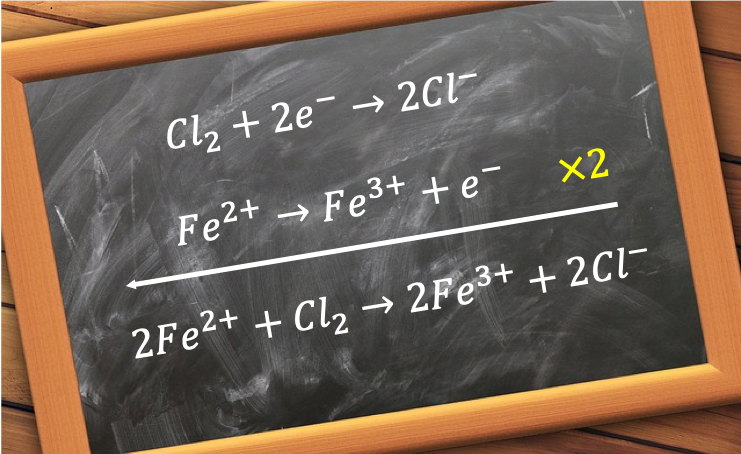
The way to construct a redox equation is to write two balanced ionic half-reaction equations, one for the oxidising agent and the other one for the reducing agent, and combine them. The method to balance each half-reaction equation is slightly different from that of a normal stoichiometric equation and involves the following steps:-
For example, the redox equation for the oxidation of acidified iron (II) sulphate by potassium dichromate (VI) is constructed as follows:
1st half-reaction equation
Fe2+ → Fe3+
Balancing the equation
Fe2+ → Fe3+ + e– eq1
(note that steps 1 through 3 and step 5 do not apply)
2nd half-reaction equation
Cr2O72- → Cr3+
Balancing the equation
Cr2O72- → 2Cr3+
Cr2O72- → 2Cr3+ + 7H2O
Cr2O72- + 14H+ → 2Cr3+ + 7H2O
Cr2O72- + 14H+ + 6e–→ 2Cr3+ + 7H2O eq2
(note that step 5 does not apply)
Next, combine the balanced half-reaction equations, cancel any common terms and ensure that all electrons are eliminated in the final equation. For this example, we multiply eq1 by 6 throughout before adding to eq2, giving:
6Fe2+(aq) + Cr2O72- (aq) + 14H+ (aq) → 6Fe3+(aq) + 2Cr3+ (aq) + 7H2O (l)

Write the redox equation for the oxidation of sodium formate by potassium manganite (VII) to form sodium carbonate and manganese dioxide in slightly basic conditions.
1st half-reaction equation
HCO2– → CO32-
Balancing the equation
HCO2– + H2O → CO32-
HCO2– + H2O → CO32- + 3H+
HCO2– + H2O → CO32- + 3H+ + 2e–
HCO2– + H2O + 3OH– → CO32- + 3H+ + 2e–+ 3OH–
HCO2– + H2O + 3OH– → CO32- + 3H2O + 2e–
HCO2– + 3OH– → CO32- + 2H2O + 2e–
2nd half-reaction equation
MnO4– → MnO2
Balancing the equation
MnO4– → MnO2 + 2H2O
MnO4– + 4H+ → MnO2 + 2H2O
MnO4– + 4H+ + 3e– → MnO2 + 2H2O
MnO4– + 4H+ + 3e– + 4OH– → MnO2 + 2H2O + 4OH–
MnO4– + 4H2O + 3e– → MnO2 + 2H2O + 4OH–
MnO4– + 2H2O + 3e– → MnO2 + 4OH–
Combining the balanced half-reaction equations, we have,
3HCO2– (aq) + OH– (aq) + 2MnO4– (aq) → 3CO32- (aq) + 2MnO2 (s) + 2H2O (l)
A disproportionation reaction is one in which an atom in a reactant undergoes both oxidation and reduction. For example,
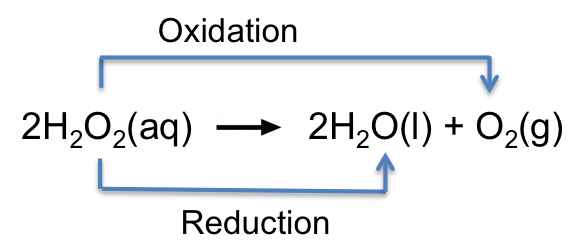
The O in H2O2 (oxidation state of O = -1) is oxidised to the O in O2 (oxidation state of O = 0) and reduced to the O in H2O (oxidation state of O = -2).
A disproportionation reaction happens when the oxidation state of an element in a compound is unstable and can achieve greater stability by splitting into two species — one at a higher oxidation state and one at a lower. This is often driven by the products having a lower total Gibbs energy than the reactant. It may also occur due to favourable changes in enthalpy and entropy.
A redox reaction is one in which oxidation and reduction occur concurrently. When two different reactants are involved, an atom, a molecule or an ion in one reactant undergoes oxidation, while an atom, a molecule or an ion in the other undergoes reduction. For example,

An oxidising agent is a species that oxidises another species and a reducing agent is a species that reduces another species. For example,

Cu2+ in the above reaction is the oxidising agent as it oxidises Zn to Zn2+ while Zn is the reducing agent as it reduces Cu2+ to Cu.
The oxidation state/number of an atom or a monatomic ion is a number assigned to the atom or monatomic ion that corresponds to its degree of oxidation.

The rules for assigning an oxidation state/number to an atom or monatomic ion is as follows:-
|
Atom |
Oxidation state/number |
|
|
Cu |
Cu |
0 |
|
Cl2 |
Cl |
0 |
|
C60 |
C |
0 |
|
Ion |
Oxidation state/number |
|
Fe3+ |
+3 |
|
Cl– |
-1 |
|
Na+ |
+1 |
|
Compound |
Atom | Assumed role |
Oxidation state/number |
|
MgCl2 |
Mg | Electron donor |
+2 |
|
CO2 |
C | Electron donor |
+4 |
|
KOH |
O | Electron acceptor |
-2 |
|
NH3 |
N | Electron acceptor |
-3 |
|
Species |
Overall charge of species |
Oxidation state/number |
|
NaCl |
0 |
+1 + (-1) = 0 |
|
NH4+ |
+1 |
-3 + (+1 x 4) = +1 |
|
PO43- |
-3 |
+5 + (-2 x 4) = -3 |

Determine the oxidation state/number of the underlined elements in AlH3, CH4 and Cr2O72-.
For AlH3, we assume that Al is the electron donor as it is more electropositive than H, and so, +3 + (H x 3) = 0. Therefore, H = -1.
For CH4, we assume that H is the electron donor as it is more electropositive than C, and so, C + (+1 x 4) = 0. Hence, C = -4.
For Cr2O72-, we assume that Cr is the electron donor as it is more electropositive than O, and so, (Cr x2) + (-2 x 7) = -2. Therefore, Cr = +6.
Oxidation is the gain in oxygen, loss of hydrogen, loss of electron or increase in oxidation number, while reduction is the loss of oxygen, gain in hydrogen, gain in electron or decrease in oxidation number.

The reactant H2 is oxidised as it gains an oxygen atom to form H2O, while O2 is reduced as it loses an oxygen to form H2O.

The reactant H2S is oxidised as it loses hydrogen atoms to form S, while Cl2 is reduced as it gains hydrogen atoms to form HCl.

The reactant Zn is oxidised as it loses two electrons to form Zn2+, while Cu2+ is reduced as it gains two electrons to form Cu.

The reactant Zn is oxidised as its oxidation state/number increases from 0 in Zn to +2 in Zn2+, while Cu2+ is reduced as its oxidation state/number decreases from +2 in Cu2+ to 0 in Cu.
2-dimensional paper chromatography is a technique in which a chromatogram is first developed using a particular solvent (either by ascending or descending paper chromatography), and then the paper is rotated 90 degrees for a second run with a different solvent.
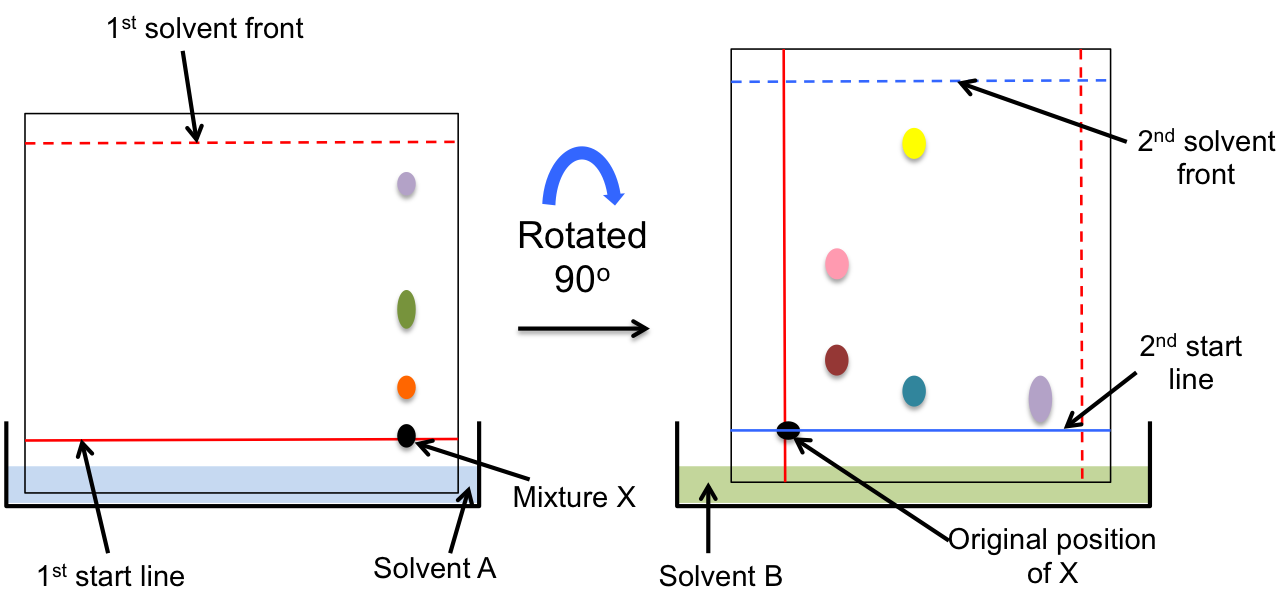
2-dimensional paper chromatography allows components that are not separated by the first solvent (due to their insolubility in that solvent) to be separated by the second solvent, thereby producing a chromatogram with better resolution than a 1-dimensional paper chromatogram.

With reference to the above diagram, what is the minimum number of chemical components in mixture X?
5