Anodising is an electrolytic process to create an inert oxide layer on a metal, which is the anode and hence the name anodising. The diagram below shows the passivation of aluminium by the formation of a layer of aluminium oxide, which protects the aluminium from corrosion. Anodised aluminium is used in many products, such as aircraft parts, consumer electronic components, and carabiners.
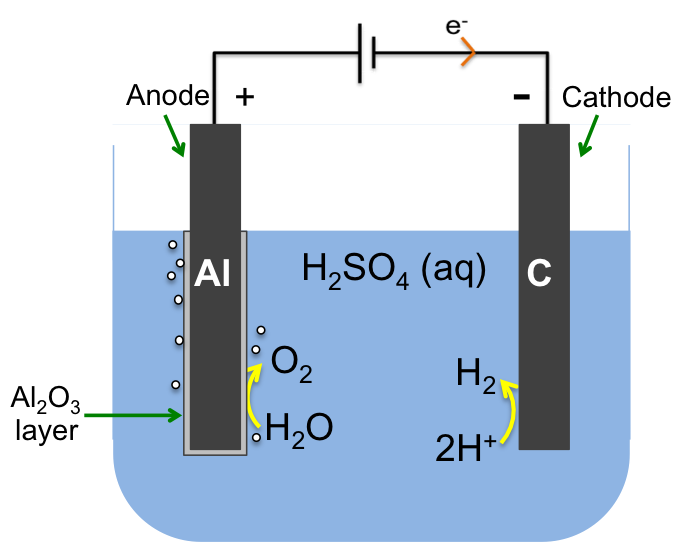
At the anode:
2H2O (l) → O2 (g) + 4H+ (aq) + 4e–
The oxygen produced reacts with the anode to form the protective layer:
3O2 (g) + 4Al (s) → 2Al2O3 (s)
with the net anodic half-cell reaction being
2Al (s) + 3H2O (l) → Al2O3 (s) + 6H+ (aq) + 6e–
Protons H+ are reduced to hydrogen gas H2 at the cathode
6H+ (aq) + 6e– → 3H2 (g)
which is typically made of carbon, and the electrolytic circuit is completed by the migration of anions such as SO42- to the anode and cations such as H+ to the cathode.
Combining the anodic and cathodic half-cell reactions gives the overall electrolytic cell reaction:
2Al (s) + 3H2O (l) → Al2O3 (s) + 3H2 (g)

Question
Will the migrating sulphate ions SO42- accumulate at the anode over time in a way that halts the electrolytic process?
Answer
Although the sulphate ions are not consumed at the anode, the evolution of oxygen gas prevents a significant concentration buildup of the ions there. In fact, a small amount of sulphate is incorporated into the growing aluminium oxide layer, and the resulting oxide film contains about 10-15% sulphate. This incorporation is a unique feature of sulphuric acid anodising and is beneficial because it makes the oxide layer more porous and easier to dye. Finally, electrical neutrality at the anode is maintained by the production of protons during the oxidation of water, which balances the negatively charged sulphate.
