A fuel cell converts chemical energy from the fuel (usually H2) to electrical energy, which is used to power engines.
With reference to the diagram below, H2 is fed into the left compartment, where it is oxidised to H+ at the anode, which is porous and impregnated with a Pt catalyst. The protons then migrate across the electrolyte (H3PO4) contained in a polymer exchange membrane that only allows the passage of H+.
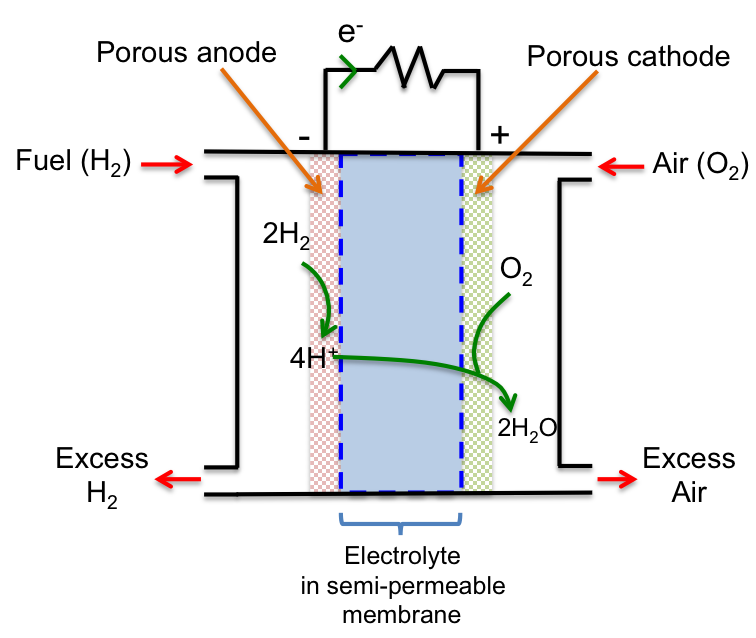
At the cathode, O2 in the air fed into the right compartment reacts with H+ and is reduced to form water. The cathode is again porous and impregnated with a Ni catalyst.
The overall redox reaction is:
2H2 (g) + O2 (g) → 2H2O (l)
Fuel cells are used to power electric motors in a variety of vehicles. They are often preferred for heavy-duty or long-range vehicles because they are lighter than batteries and can be refuelled in minutes. For example, cars, forklifts, buses, passenger trains and even submarines are powered by fuel cells. They also provide electricity for onboard systems in space shuttles. In fact, the byproduct water is so pure that it serves as drinking water for astronauts. Finally, the compact design of fuel cells allows them to function as portable power packs for specialised applications, such as for soldiers in the field.
next article: The alkaline battery
Previous article: Other forms of electrochemical cell
Content page of basic electrochemistry
Content page of Basic chemistry
Main content page