Molecular rotational energies are quantised states arising from rotation about principal axes, determined by the moments of inertia. These levels are fundamental in molecular spectroscopy.
Diatomic rigid rotor
For a diatomic rigid rotor, the rotational Hamiltonian is given by eq4a, . Here,
is the square of the total angular momentum operator, with the eigenvalues
(derived using orbital angular momentum ladder operators), where
is the rotational quantum number and
. Therefore, the rotational energy levels of a diatomic molecule are given by:
Eq44 can also be expressed in terms of the rotational constant :
where is the Planck constant and
is the speed of light.
However, rotational energies are typically reported as wavenumbers in molecular spectroscopy. Substituting and
into eq45 yields
, which is a function of
. Therefore,
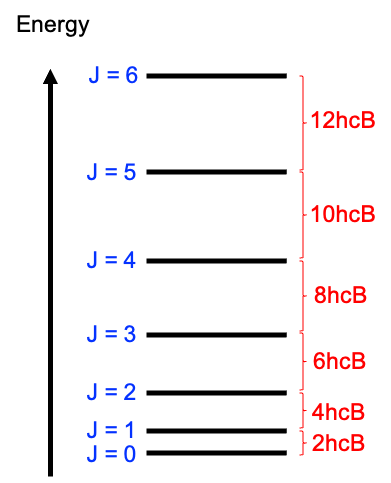
This indicates that the separation between adjacent rotational levels increases linearly with (see above diagram), as given by:
Furthermore, each energy level (i.e. eq44 for a particular value of ) is
-fold degenerate due to the allowed eigenvalues of the
–component of angular momentum, which are
, where
. In the absence of an external magnetic field, these sublevels are degenerate.
Other linear rotors
A rigid linear triatomic molecule (e.g. HCN) behaves similarly as a diatomic rigid rotor when rotating about an axis perpendicular to its bond axis. The moment of inertia about the molecular axis is essentially zero due to the absence of perpendicular mass displacement. Therefore, the rotational energy levels are, per eq44:
where is given by eq15.
Similar to diatomic molecules, each is
-fold degenerate in the absence of an external magnetic field.

Question
Is C2H2 also a linear rotor?
Answer
Yes. Its rotational energy levels are also given by eq44 and eq46.
Symmetric rotors
Symmetric rotors have two equal moments of inertia about mutually orthogonal axes. Each of these is perpendicular to a third rotational axis associated with a distinct, non-zero moment of inertia
. If
, the molecule is known as a prolate symmetric rotor (shaped like a cigar) and rotates more easily around the principal axis. Examples include NH3 and CHCl3. If
, the molecule is called an oblate symmetric rotor (flattened like a disc), and it rotates more easily around an axis perpendicular to the disc. Examples include C6H6 and BF3.
The rotational Hamiltonian of a rigid symmetric rotor in the molecule-fixed frame is given by eq4d (by replacing the orthogonal lab-frame axes with orthogonal molecular axes
):
Conventionally, and
. Furthermore, from eq75,
. Therefore,
With reference to eq132, and replacing the notations ,
and
with
,
and
, where
, we have
. It follows that the rotational energy levels of a rigid symmetric rotor are given by:

Question
Is a quantum number? Is it the same as
(or
)?
Answer
In eq48, refers to the component of the angular momentum operator along the symmetry axis (or
-axis) in the molecule-fixed frame, not the
-axis in the laboratory frame. Although its eigenvalues have the same form as those of
in the laboratory frame, it is a different quantum number from
(or
). An analogy can be made with the projections of the spin vector of a spinning top: its projection along its own axis differs in general from the projection along an arbitrary laboratory axis, unless the axes are aligned. While both projections can take similar ranges of values (assuming these values are quantised), they are not equal in general. Likewise,
and
are related to projections in different frames and are different quantum numbers.
Substituting and
into eq49 yields:
Substituting and
into eq50 gives
, which is a function of two rotational quantum numbers
and
. Therefore,
When , there is only one distinct moment of inertia, with eq51 reducing to eq46, where
. In this case, each
is
-fold degenerate in the absence of an external magnetic field. If
,
is determined by the magnitudes of
and
. Since
is the same for
and
for each
in eq49, the energy levels for
and
are degenerate. Additionally, there are
possible
substates for each value of
. Therefore, the total degeneracy when
is
in the absence of an external magnetic field.
Spherical rotors
A spherical rotor has three equal moments of inertia about three mutually orthogonal axes. It belongs to a point group that is a parent group of that of a symmetric rotor. Therefore, the rotational energy levels of a spherical rotor can be described by eq49 with
, which is equivalent the condition
in eq50. This implies that
remains a function of both
and
, and that for a given energy level,
is not restricted to just two values (as in the case of a symmetric rotor), but can take on
different values. It follows that each energy level is
-fold degenerate in the absence of an external magnetic field.