The Pauling electronegativity scale is a dimensionless numerical system that ranks atoms according to their ability to attract shared electrons in a chemical bond.
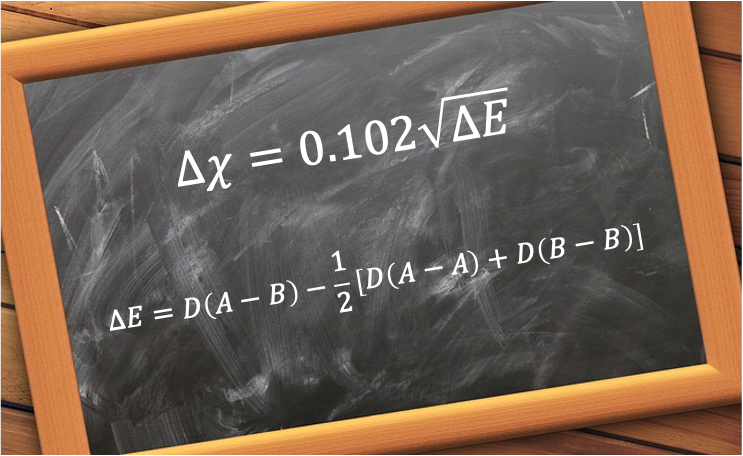
When two atoms form a bond, they do not always share electrons equally. Instead, one atom may attract the bonding electrons more strongly than the other, creating a polar bond. Linus Pauling proposed in 1932 that this unequal sharing could be quantified by assigning each element a relative value known as its electronegativity .
-
- Atoms with high
(like fluorine) strongly attract electrons.
- Atoms with low
(like alkali metals) attract electrons weakly.
- Atoms with high
Pauling’s approach was based on bond energies. Specifically, he noticed that the experimental bond dissociation energy between two different atoms (A–B) is often greater than expected compared to the average bond dissociation energies of A–A and B–B bonds. He attributed this “extra” strength to the ionic character of the bond and defined the extra stabilisation energy as:
where is the bond dissociation energy of the molecule X-Y.
Working empirically with the bond dissociation energies of different molecules in electronvolts (eV), Pauling was able to create a linear scale by taking the square root of , which he related to the difference in electronegativity between atoms A and B:
For a homonuclear diatomic molecule A-A, . If the bond dissociation energies are expressed in kJ/mol, the formula becomes:
where 1 eV = 96.48 kJ/mol and .
Because the formula only calculates the difference in electronegativity, the scale needs an arbitrary starting point. Pauling originally set hydrogen to 2.1 to ensure that most elements had positive values. Once the scale was anchored to hydrogen, the value for fluorine was calculated to be approximately 3.98 (often rounded to 4.0 in textbooks).
The difference in electronegativity between two bonded atoms helps predict whether a bond is nonpolar covalent, polar covalent or ionic. As a rule of thumb,
-
- Small difference (≈ 0–0.4): nonpolar covalent, e.g. H-H (
).
- Moderate difference (≈ 0.5–1.7): polar covalent, e.g. H-Cl (
).
- Large difference (> 1.7): ionic, e.g. Na-Cl (
).
- Small difference (≈ 0–0.4): nonpolar covalent, e.g. H-H (

Question
Deduce if water has an electric dipole given and
.
Answer
Since , the O-H bond is polar covalent. The electrons are not shared equally, with the more electronegative oxygen pulling the shared electrons closer to itself. This creates an electric dipole, where the oxygen atom has a partial negative charge and the hydrogen atoms have partial positive charges. Such polarity allows water molecules to form hydrogen bonds, giving water unique properties such as a high boiling point.