The inductive effect is the shift of electron density in -bonds caused by electron-withdrawing or electron-donating groups, resulting in polarised bonds.
This effect occurs because different atoms or groups influence electron density according to their electronegativity relative to the atoms to which they are bonded. Atoms or groups that are more electronegative tend to withdraw electron density, while those that are less electronegative can donate electron density through the -bond framework. In organic chemistry, the inductive effect is often discussed for substituents attached to carbon chains, where electron density is shifted along the bonds. As a result, atoms near an electron-withdrawing group may become slightly electron-deficient and develop a partial positive charge, whereas atoms near an electron-donating group may become slightly electron-rich and develop a partial negative charge. Whether a substituent is labelled electron-withdrawing (–I) or electron-donating (+I) depends entirely on how its electronegativity compares to a hydrogen atom bonded to that same position.
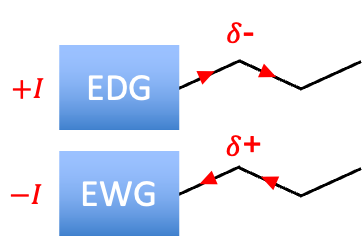
Electron-withdrawing groups (-I)
To quantum-mechanically analyse the inductive effect caused by electron-withdrawing substituents (classified as –I substituents), we consider an atom in which the energy of an electron can be expressed as two main contributions:
where is the Hamiltonian,
is the kinetic energy of the electron and
is the electrostatic potential energy arising from attraction to the nucleus.
The potential energy is approximately
where is the effective nuclear charge and
is the electron-nucleus distance.
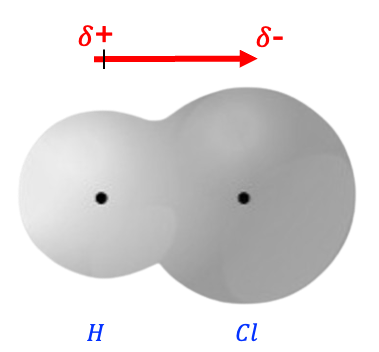
Consider the heteronuclear diatomic molecule HCl, in which the Cl atom is more electronegative than the H atom. Since , the potential energy of an electron on Cl is more negative than that on H, lowering the valence orbital energy of Cl relative to that of H. In other words, the more electronegative atom or group usually has a lower-energy valence orbital.
When the two atoms combine to form a -bond, the bonding molecular orbital (MO) wavefunction
is
where and
are the atomic orbital (AO) wavefunctions of H and Cl respectively, and
and
are real-valued coefficients that are key to explaining the inductive effect.
If is normalised and the AOs are orthogonal, the electron density of the
-bond is given by
This implies that and
determine the contribution of each AO to the electron density of the bond. The larger coefficient corresponds to a greater contribution and therefore greater electron density in the region of that atom. Because Cl is more electronegative, we expect
. To justify this, we multiply the eigenvalue equation
on the left by
and integrate to give
where ,
,
and we have
-
- used the Hermitian property of the Hamiltonian
- assumed that the set
is orthonormal and expectation values are real, so that
.
- used the Hermitian property of the Hamiltonian
Applying the variational principle to eq380 by setting the partial derivative of with respect to each coefficient to zero, we obtain a set of simultaneous equations known as secular equations:
Expressing eq382 and eq383 in matrix form yields:
Eq384 is a linear homogeneous equation with non-trivial solutions only if
Expanding the determinant gives the characteristic equation:
with two solutions corresponding to the bonding MO () and antibonding MO (
):
The energy separation between these two MOs is
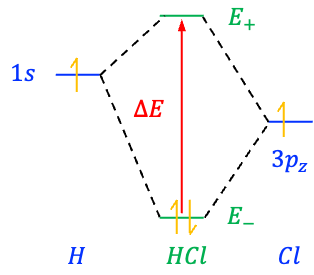
If , then
, which implies that the two AOs do not interact and no splitting occurs. It follows that
increases the separation between the two MOs, with a larger
resulting in a greater separation, and hence, a greater stabilisation of the bonding MO.
Rearranging eq383 and taking absolute values on both sides yield:
Substituting in eq385 into
gives:
Multiplying and dividing the RHS by its conjugate yields
Since , then
.
is the expectation value of the hydrogen AO, i.e.
, which is less negative than
. So,
and
. Therefore,
, where
, or equivalently,
Substituting this into eq386 results in
This proves that the coefficient of the more electronegative atom corresponds to a greater contribution to the -bond, and therefore, greater electron density in the region of that atom.
In terms of polyatomic aliphatic molecules, the inductive effect is transmitted along covalent bonds in the chain, but its strength decreases rapidly with distance. To illustrate this, consider the C-C-Cl -framework in the molecule CH3CH2Cl, with the following bonding MO wavefunction:
where 1 denotes the terminal carbon, 2 denotes the carbon bonded to Cl and 3 denotes the chlorine atom.
The difference in electronegativity between chlorine and the adjacent carbon is significantly larger than that between the two carbon atoms. We would therefore expect , with the electron density skewed towards chlorine, leaving the middle carbon slightly electron-deficient and effectively more electronegative relative to the terminal carbon atom. Applying similar reasoning to the C-C bond, we obtain
, and consequently,
This result implies that the inductive effect transmitted along covalent bonds in a chain decreases in strength with increasing distance from the substituent. It also means that the C-Cl moiety may be regarded as an electron withdrawing group. More generally, a group containing several atoms may behave collectively as an electron-withdrawing group if its internal bonding renders an atom electron-deficient relative to the surrounding network and capable of attracting electron density from neighbouring bonds.
For example, in the formyl group (–CHO), the carbonyl oxygen strongly stabilises the system of the C=O bond, lowering the energy of orbitals centred on the carbonyl carbon. As a result, the carbonyl carbon becomes relatively electron-deficient and can draw electron density from the neighbouring
framework. Such redistribution of electron density can significantly influence the chemical behaviour of molecules. For instance, inductive effects can alter the acidity of different amino acids by stabilising or destabilising charged intermediates, and can also affect reaction mechanisms by increasing the electrophilicity of certain atoms, making them more susceptible to nucleophilic attack.
Electron-donating groups (+I)
A group that has the opposite effect on a chain of atoms compared to an electron-withdrawing group is known as an electron-donating group.
The same molecular orbital reasoning can be used to explain electron-donating groups. The key idea is that the direction of electron density shift depends on the relative energies of the interacting atomic orbitals, which are reflected in the Coulomb integrals . For an electron-donating group, the situation is reversed. The atom attached to the carbon chain has higher-energy valence orbitals, corresponding to a larger Coulomb integral than that of the neighbouring carbon atom. When the bonding molecular orbital is formed, the coefficient on this substituent becomes relatively smaller than that on the adjacent carbon atom. Consequently, electron density is pushed into the carbon skeleton. Thus, the substituent effectively donates electron density through the
framework, producing a positive inductive effect (+I).
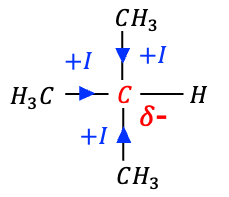
Alkyl substituents provide a simple example. Because carbon is slightly more electronegative than hydrogen, the electron density in C-H bonds of an alkyl group such as CH3– is shifted towards the carbon atom. This leaves the carbon atom relatively electron-rich and its valence orbitals at slightly higher energy than those of a methylene carbon (CH2). When these units combine to form the ethyl group CH3CH2-, the methyl fragment behaves as an electron-donating substituent. Comparing H3C-CH2– and H-CH2-, we expect the methylene carbon in the ethyl group to be more electron-rich than that in the methyl group, and hence the ethyl group to be a stronger electron-donating group than the methyl group. It follows that, in terms of increasing +I effect: