The quadratic Stark effect is the shift of atomic or molecular energy levels proportional to the square of an applied electric field, arising when non-degenerate states produce only second-order energy corrections.
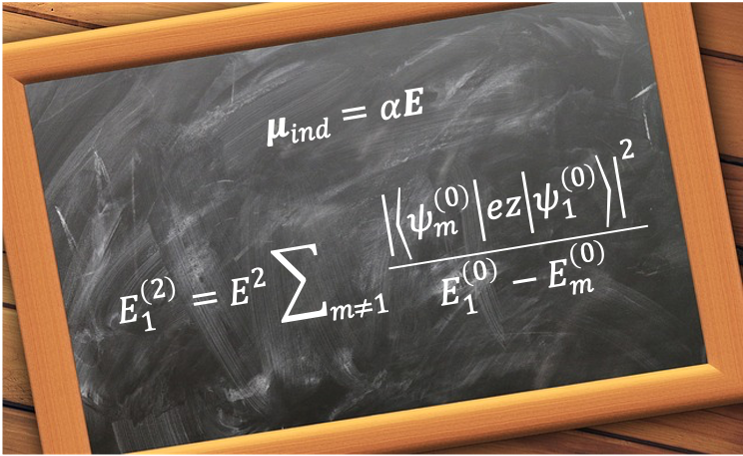
As explained in the previous article, the ground state () of hydrogen does not exhibit a linear Stark effect, which is defined as the first-order energy correction in perturbation theory, because its spherical symmetry implies that it possesses no permanent electric dipole moment. With reference to eq270a, the second-order energy correction for the ground state of hydrogen
is
where
is the perturbing part of the Hamiltonian due to the Stark effect (see eq362)
is the external electric field directed along the
-axis
is a linear combination (mixing) of excited eigenstates, with
given by eq269a
and
are the ground state eigenfunction and eigenvalue
and
are the eigenfunctions and eigenvalues for
.
Since corresponds to the 1s wavefunction, which is an even function under spatial inversion, and
is odd under spatial inversion,
is an odd function. Consequently, for
,
must be odd. The term in the summation with the smallest magnitude of the denominator
contributes most significantly to
. This occurs when
, implying that the 2p wavefunctions
(which are the only odd-parity states in the
level) dominate the correction to the eigenvalue.
It follows that the ground state of hydrogen exhibits a Stark effect when the second-order energy correction is taken into account. To show that it is a quadratic effect and that the associated electric dipole moment is an induced dipole moment, we substitute eq362 into eq370 to obtain
Clearly, the second-order energy correction is proportional to , and hence represents a quadratic Stark effect.
In classical electromagnetism, the energy of an electric dipole moment in an external field is given by eq354, or in differential form:
Substituting the definition of an induced electric dipole moment into
gives
Since the external electric field is directed along the -axis,
If in eq371 is an energy shift arising from an induced dipole moment, it must have the form of given in eq372. Comparing eq371 with eq372 yields
where is the polarisability of the atom, which is a measure of the degree to which the electron in hydrogen can be displaced relative to the nucleus.
Therefore, in eq371 is an energy shift arising from an induced dipole moment. Even though the ground state of hydrogen does not possess a permanent dipole moment, it exhibits a quadratic Stark effect due to the electric dipole moment induced by an external electric field.