Lithium-ion batteries power everything from smartphones and laptops to electric vehicles and energy storage systems. They are efficient, lightweight and rechargeable. However, under certain conditions, they can overheat, expand and even catch fire. Understanding why this happens comes down to how these batteries are built and what can go wrong inside them.
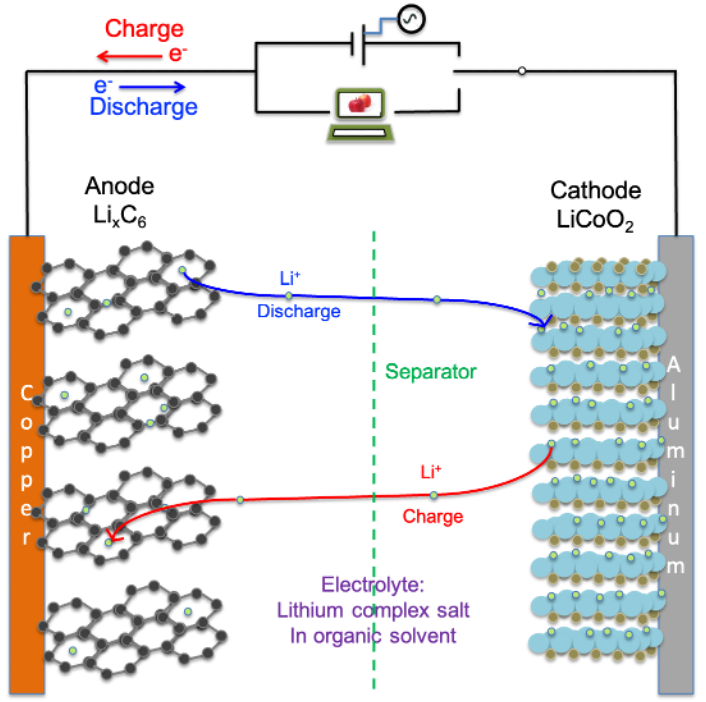
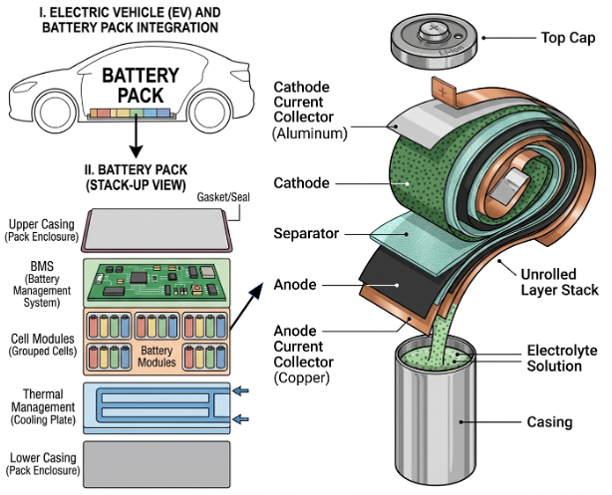
A lithium-ion battery consists of three key components: anode, cathode and the electrolyte (see diagram above). Separating the anode and cathode is a thin porous membrane called the separator, which plays the critical role of keeping the two electrodes apart while still allowing ions to pass through. As the battery charges and discharges, lithium ions shuttle back and forth between the electrodes, storing and releasing energy.
Under normal conditions, lithium-ion batteries are safe. Problems arise when internal or external factors disrupt their delicate balance, with the most serious outcome being thermal runaway — a chain reaction in which rising temperature accelerates further heat generation. The main trigger for a thermal runaway is an internal short circuit, which occurs when the anode and cathode come into direct contact. When this happens, charges no longer flow through the higher-resistance electrolyte, but instead travel via a very low-resistance path between the electrodes. This surge in internal current flow results in rapid heat generation that can lead to combustion.
So what causes the anode and cathode to come into direct contact? Physical damage, such as dropping, crushing or puncturing a battery can deform its internal structures. Manufacturing defects may leave contaminants, like metal particles or dust, inside the battery that can pierce the separator. Another cause is lithium plating, which can lead to dendrite growth.
Lithium plating occurs when lithium ions deposit as metallic lithium on the surface of the anode, Li+(aq) + e– → Li(s), instead of moving into the anode’s internal structure (a process known as intercalation). Fast charging drives lithium ions rapidly towards the anode and can overload its surface. If the intercalation rate is slower than the incoming flux, the excess ions tend to accumulate and are reduced to metallic lithium on the anode surface. Low operating temperatures (< 273.15 K), which increase the viscosity of the electrolyte and decrease ionic mobility, can also promote lithium plating (the slower diffusion of ions into the graphite layers causes them to accumulate and plate onto the surface).
Initially, this plated lithium layer is often relatively uniform, so the anode and cathode remain physically separated by the separator, although the battery capacity may be affected. However, if the plated lithium grows unevenly over time, it can form needle-like structures called dendrites. These body-centred cubic structures can pierce the separator and reach the cathode, creating a direct, low-resistance path between the electrodes and leading to an internal short circuit.
Finally, gas buildup in a lithium-ion battery, caused by electrolyte decomposition over time, can also result in thermal runaway. This decomposition involves the battery’s electrolyte, which typically consists of the lithium salt LiPF₆ dissolved in an organic solvent such as ethylene carbonate or dimethyl carbonate. Under stress (e.g. overcharging or high temperature), LiPF₆ can decompose to PF₅ (LiPF₆ → LiF + PF₅), which then reacts with trace amounts of water to produce hydrogen fluoride gas (PF5 + 4H2O → H3PO4 + 5HF). The organic solvents can also oxidise or decompose, producing gases like CO, CO₂ or hydrocarbons, leading to battery swelling commonly observed as bulging laptop casings. The increased internal pressure may displace the electrodes, causing an internal short circuit, or even rupture the battery. This significantly increases the risk of explosion, especially if the gases are flammable.
To mitigate such risks, high-quality lithium-ion battery designs use a combination of materials, architecture and protective systems to prevent lithium plating, dendrite formation, gas buildup, and short circuits. For instance, high-quality graphite, silicon-graphite composites or lithium titanate anodes ensure even lithium intercalation, reducing the risk of plating. Additives are also included in the electrolyte to regulate lithium deposition and suppress dendrite growth. Furthermore, multi-layer microporous polymer separators can resist puncture by dendrites.
Additionally, devices powered by lithium-ion batteries incorporate electronic circuitry that monitor battery voltage, current and temperature (battery management systems) to prevent overcharging and over-discharging. Cooling systems, such as personal computer fans, also help reduce overheating and electrolyte decomposition.

Question
Do electric cars (EVs) also use lithium-ion batteries? If so, why do they rarely catch fire?
Answer
Yes, most EVs use lithium-ion batteries. While the chemistry is similar to that of batteries in phones or laptops, EV battery systems are much more advanced, with high-quality electrodes and robust casings that protect against physical damage. Sophisticated battery management and thermal management systems are also employed to prevent overheating (see diagram below).
In summary, lithium-ion batteries catch fire not because they are inherently unsafe, but because their high energy density makes them sensitive to internal failures and external stress. When key safeguards (separator, electrolyte stability or controlled ion movement) are compromised, a cascade of reactions can lead to thermal runaway. However, with continued advances in materials science, battery design and management systems, the risks are being steadily reduced. When properly designed, manufactured and used, lithium-ion batteries remain a safe and indispensable technology in modern life.