The pKa of an amino acid is the specific pH at which one of its ionisable groups is 50% protonated.
This concept is central to understanding how amino acids behave in different chemical and biological environments. Because amino acids contain multiple ionisable groups (typically an amino group, a carboxyl group and sometimes an ionisable side chain), their charge state can change depending on the surrounding pH. This directly affects properties such as solubility, structure, and interactions with other molecules, especially in proteins where even small shifts in charge can alter folding or function.

Question
Why is the pKa of an amino acid not defined as the negative logarithm of the acid dissociation constant for a specific ionisable group?
Answer
Both definitions are equivalent. The formal definition is . For the equilibrium
,
which rearranges to the Henderson-Hasselbalch equation:
When one ionisable group is 50% protonated, , so,
. Thus, defining pKa as the pH at which a group is 50% protonated is simply a practical interpretation of the mathematical definition. It is easier to determine this point experimentally from titration curves (the buffering midpoint) than to directly measure
, especially for amino acids with multiple ionisable groups.
Several factors influence the pKa values of amino acids, including:
-
- Resonance stabilisation.
- Inductive effects.
- Intramolecular hydrogen bonding.
- The local environment surrounding the amino acid.
Resonance stabilisation
Functional groups that can delocalise electrons in the conjugate base lower pKa values. In other words, the more stable the conjugate base, the stronger the acid, and thus the lower its pKa. Carboxyl groups generally have lower pKa values (more acidic) than amino groups because their negative charge, once deprotonated, is stabilised by resonance. In contrast, amino groups tend to hold onto their protons more strongly, resulting in higher pKa values.
For example, when the phenolic group of tyrosine loses a proton (pKa ≈ 10.46), the negative charge is delocalised into the benzene ring and the conjugate base is resonance-stabilised. In comparison, the pKa of cyclohexanol is about 16 (negative charge of the conjugate base is not delocalised and remains on the oxygen atom).
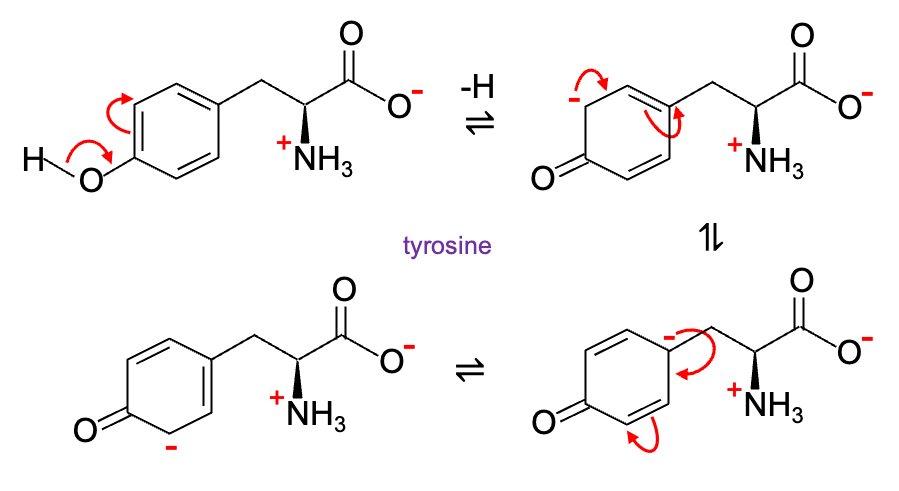
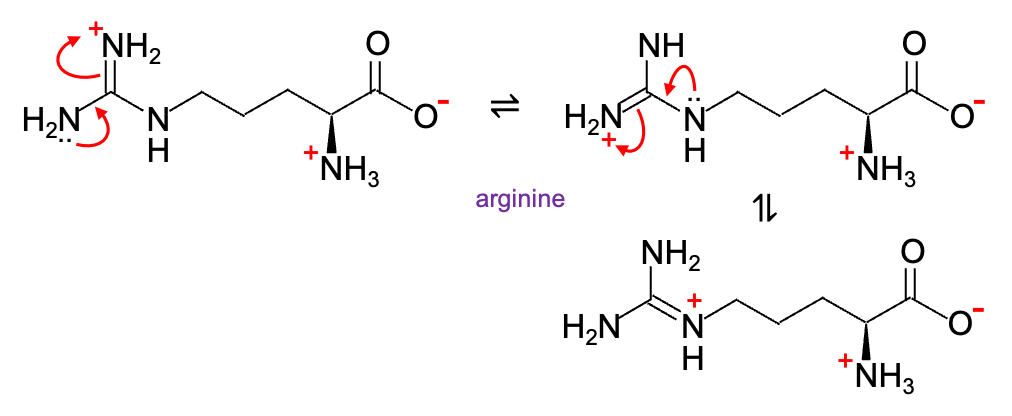
Resonance stabilisation also applies to the amine group of an amino acid, increasing its pKa value and making it a strong base. For instance, the conjugate acid of arginine is stabilised (pKa ≈ 12.48) when its guanidino group is protonated (c.f. lysine pKa ≈ 10.54).
Inductive effects
An electron-withdrawing group stabilises the negative charge formed after deprotonation of a carboxyl group in an amino acid, thereby lowering its pKa. Conversely, an electron-donating group destabilises the charged form and increases the pKa.
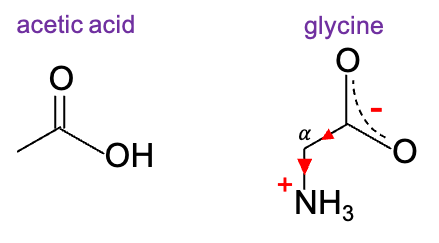
For example, the carboxyl group of glycine is more acidic (pKa ≈ 2.35) than acetic acid (pKa ≈ 4.76) because its conjugate base is stabilised by both inductive and resonance effects. Notably, the amino group, which is protonated under physiological conditions (pH ≈ 7.4), acts as an electron-withdrawing group.
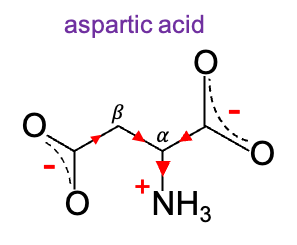
Another example is aspartic acid. Since the –I effect decreases with distance, the -carboxylate group (pKa ≈ 1.99) is stabilised more strongly than the
-carboxylate group (pKa ≈ 3.90). Parenthetically, aspartic acid residues in proteins are usually negatively charged at physiological pH, contributing to electrostatic interactions such as salt bridges with positively charged amino acids (e.g. lysine or arginine). This helps to stabilise protein structure.
On the other hand, an alkyl side chain exerts a +I effect on other functional groups in an amino acid. For example, the methyl group in alanine acts as an electron-donating group and increases the electron density on the nitrogen atom of the adjacent amino group, making it a slightly stronger base (pKa ≈ 9.87) than that of glycine (pKa ≈ 9.78).


Question
Is the side-chain carboxyl group of aspartic acid more or less acidic than that of glutamic acid?
Answer
It is more acidic because the shorter carbon chain in aspartic acid places the side-chain carboxyl group closer to the electron-withdrawing amino group, making it easier to lose a proton.
Intramolecular hydrogen bonding
Additionally, intramolecular hydrogen bonding can influence pKa by stabilising the deprotonated state, lowering the pKa of the carboxyl group. The hydroxyl group in the side chain of serine may form an intramolecular hydrogen bond with the carboxylate oxygen. This also occurs in threonine, whose pKa (about 2.09) is slightly lower than that of serine (pKa ≈ 2.19). While the relatively small substituents attached to the -carbon in serine allow the Ca-Cb bond to rotate more freely, the bulky methyl group in threonine reduces steric hindrance by favouring a gauche conformation, in which the hydroxyl group is positioned closer to the carboxyl group, increasing the probability of forming an intramolecular hydrogen bond. This makes threonine slightly more acidic than serine.
