Ortho/para-directing and meta-directing groups are substituents that guide incoming electrophiles to the ortho and para positions or to the meta position, respectively, on an aromatic ring.
This directing effect arises from how the substituent interacts with the ring’s electron density. Ortho/para-directing groups are typically electron-donating (e.g. –OH, –NH₂, –CH₃); they increase electron density at the ortho and para positions through resonance or inductive effects, stabilising the intermediate carbocation during electrophilic aromatic substitution. In contrast, meta-directing groups are usually electron-withdrawing (e.g., –NO₂, –COOH, –CN); they pull electron density away from the ring, making the ortho and para positions less stable for substitution and thus favouring attack at the meta position.
Ortho/para-directing groups
A classic example of an ortho/para-directing group is the hydroxyl group in phenol. Although it is inductively withdrawing (-I) due to the electronegativity of the oxygen atom, the lone pair of electrons on oxygen exerts a +M effect on the benzene ring, overwhelming the inductive effect and activating the ortho and para positions for electrophilic substitution.
To quantum-mechanically analyse the mesomeric effect of the hydroxyl group, we consider the general molecular orbital (MO) wavefunction of phenol:
where and
represent the coefficients and the p atomic orbital wavefunctions respectively;
corresponds to the oxygen atom,
corresponds to the substituted carbon, and
through
correspond to the remaining carbons in an anticlockwise direction.
Applying the Hückel method gives the following secular determinant:
where
is the Coulomb integral for oxygen, with
being a factor that lowers the orbital energy.
is the resonance integral for overlap between C and O.
is the Coulomb integral for carbon atoms.
is the resonance integral between carbon atoms.
is the energy of an MO.
Incidentally, the minor of the above secular determinant is the secular determinant of benzene.
The next step involves estimating trial values for and
. Since oxygen is more electronegative than carbon, we set
, lowering the MO relative to carbon. Furthermore, we set
, which is lower than the reference of
for every resonance integral between carbon atoms, because the oxygen’s 2p orbitals are more contracted than carbon’s, leading to a less effective overlap between the p orbitals of oxygen and carbon versus that between two carbon atoms.
Using the matrix identity of , and multiplying both sides of the secular determinant equation by
, give
where .
Expanding the determinant yields the characteristic equation:
with solutions
Substituting the solutions back into yields:
| MO | Type | ||
| 1.994 | Antibonding | ||
| 1.192 | Antibonding | ||
| 0.617 | LUMO | ||
| -0.213 | HOMO | ||
| -0.744 | Bonding | ||
| -1.419 | Bonding | ||
| -2.427 | Bonding |
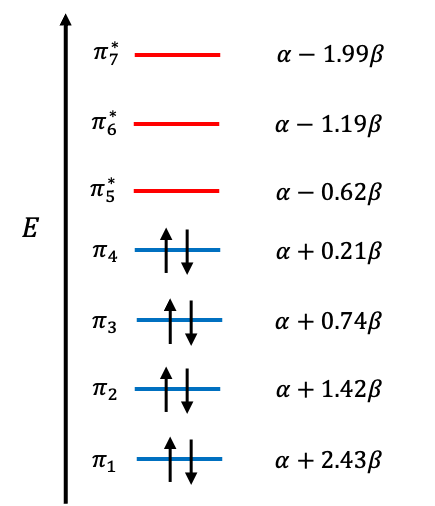
The reactivity of phenol is reflected in its HOMO energy level, , which is higher than that of benzene (
). Because the HOMO is higher in energy, the electrons are less tightly held and more nucleophilic than those in benzene towards electrophiles. To further understand the directing effects of the hydroxyl group, we determine the coefficients of the HOMO wavefunction as follows:
which implies:
Using an optimising tool, such as Microsoft Excel Solver, the normalised HOMO coefficients are approximately:
| Coefficient | (HOMO) | HOMO electron density |
| c0 | 0.623 | 0.777 |
| c1 (ipso) | -0.323 | 0.208 |
| c2 (ortho) | -0.344 | 0.236 |
| c3 (meta) | 0.121 | 0.030 |
| c4 (para) | 0.415 | 0.344 |
| c5 (meta) | 0.121 | 0.030 |
| c6 (ortho) | -0.344 | 0.236 |
where the HOMO electron density (see diagram below) is given by for two electrons.

The calculated values show that the ortho and para carbons have higher electron densities than the meta carbons, with the para carbon having the highest. This is due to the +M and –I effects of the hydroxyl group. When phenol reacts with an electrophile such as bromine at low temperatures in non-polar solvents (e.g. CS2 or CCl4), mono-brominated products are formed, giving a para-bromophenol to ortho-bromophenol ratio of about 80:20. At higher temperatures, phenol reacts with aqueous bromine to yield the fully substituted 2,4,6-tribromophenol. Therefore, the hydroxyl group is known as an ortho/para-directing group. Substituents with similar effects include the amino group (-NH2), substituted amino groups (-NR2), alkoxy groups (-OR), phosphino and thio groups (-PR2 and -SR) and the phenyl group (-C6H5).
Meta-directing groups
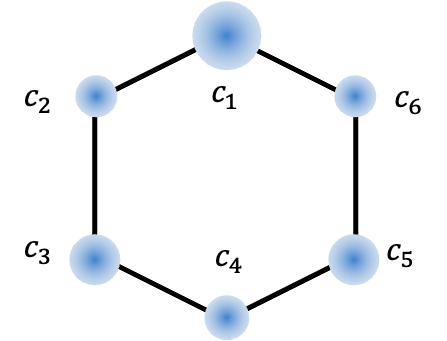
An example of a meta-directing group is the ammonium group -NH3+ in the anilinium ion C6H5-NH3+. This substituent is purely inductively withdrawing (-I) due to the positively charged nitrogen atom. To analyse the inductive effect of the ammonium group quantum mechanically, we consider the general molecular orbital (MO) wavefunction of the anilinium system:
where and
represent the coefficients and the p atomic orbital wavefunctions respectively;
corresponds to the substituted carbon, and
through
correspond to the remaining carbons in an anticlockwise direction.
As the nitrogen atom lacks a lone pair of electrons to interact with the benzene π-system, the substituted carbon atom is treated as being perturbed by the electron-withdrawing group, leading to the following secular determinant:
Expanding the determinant yields the characteristic equation:
with solutions
Substituting the solutions back into yields:
| MO | Type | ||
| 1.891 | Antibonding | ||
| 1.000 | Antibonding | ||
| 0.705 | LUMO | ||
| -1.000 | HOMO | ||
| -1.317 | Bonding | ||
| -2.278 | Bonding |
To further understand the directing effects of the ammonium group, we determine the coefficients of the HOMO wavefunction as follows:
Solving the above six simultaneous equations, along with the normalisation equation, using algebra for the HOMO coefficients and Excel Solver for the remaining coefficients, give:
| Coefficient | Electron density | |||
| c1 (ipso) | 0.646 | -0.517 | 0.000 | 1.370 |
| c2 (ortho) | 0.413 | -0.082 | 0.500 | 0.855 |
| c3 (meta) | 0.295 | 0.409 | 0.500 | 1.008 |
| c4 (para) | 0.259 | 0.621 | 0.000 | 0.904 |
| c5 (meta) | 0.295 | 0.409 | -0.500 | 1.008 |
| c6 (ortho) | 0.413 | -0.082 | -0.500 | 0.855 |
where the electron density is given by for two electrons in each MO.
The calculated values show that the meta carbons have higher electron densities than the ortho and para carbons. When the anilinium ion reacts with an electrophile such as the nitronium ion NO2+ at low temperatures, m-anilinium is the major product. Therefore, the ammonium group is known as a meta-directing group. Substituents with similar effects include the nitro group (-NO2), sulfonyl groups (-SO2R), the cyano group (-CN), formyl and acyl groups (-CHO and -COR) and the carboxyl group (-CO2H).

Question
Why doesn’t the electrophile attack the ipso (latin for “itself” or “that very one”) carbon and displace the ammonium group?
Answer
Although the Hückel method indicates that the ipso carbon has the highest electron density, it considers only the π-electrons and does not account for constraints imposed by the σ-framework. Electrophilic attack at the ipso carbon would require either displacement of the ammonium group or formation of a stable σ-complex. Both pathways are energetically very unfavourable because -NH3+ is a poor leaving group and the ipso carbon has already reached its maximum valency.