What is the formula describing the buffer capacity of water?
Consider a solution containing water and a strong base with the following equilibria:
With reference to the above equilibria, the sum of the number of moles of H+ and Na+ must equal to that of OH– (see this article for details). As the volume is of the solution is common to all ions,
Substituting cb = [Na+] and Kw = [H+][OH–] in eq3
Substitute eq1 and pH = –log[H+] in eq4
To determine the maximum or minimum buffer capacity of water,
Let
Substitute Kw = [H+][OH–] in the above equation
This means that a stationary point for the function in eq5 occurs when [OH–] = [H+] , i.e. at pH = 7. Let’s investigate the nature of this stationary point by differentiating eq6.
Substitute Kw = [H+][OH–] in the above equation, noting that [OH–] = [H+]
If we repeat the above steps and consider the case of the addition of a strong acid to water, we end up with the same conclusion. Hence, water has minimum buffer capacity at pH = 7.
The buffer capacity of water over the entire range of pH can be found by substituting [H+] = 10-pH in eq5 to give
and plotting the above equation with βw as the vertical axis and pH as the horizontal axis to give:
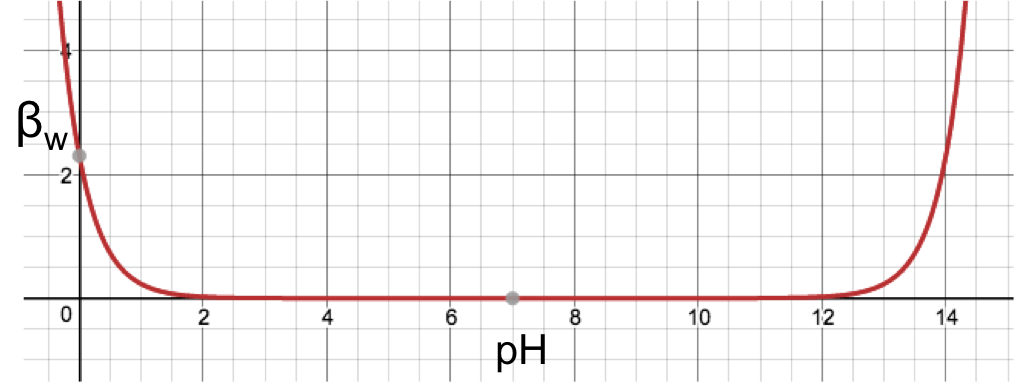
From the graph, the buffering capacity of water increases infinitesimally from a minimum of 0 at pH 7, to 0.023 at pH 2 on the left and pH 12 on the right, and then drastically to 2.303 at pH 0 on the left and pH 14 on the right. This means that water is only relatively effective as a buffer at extreme pH levels.
