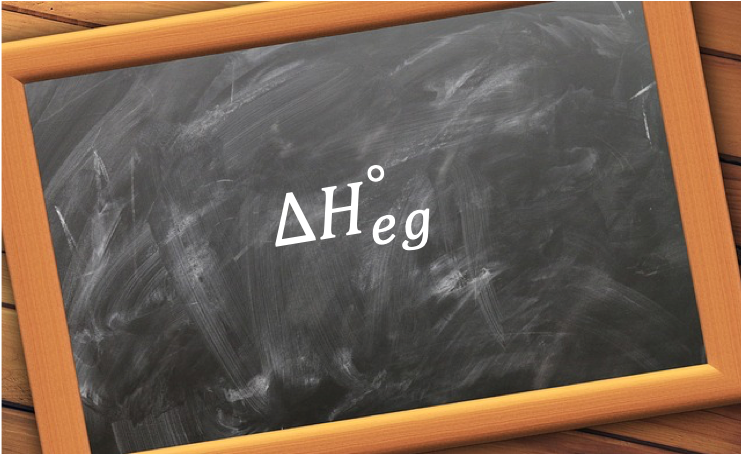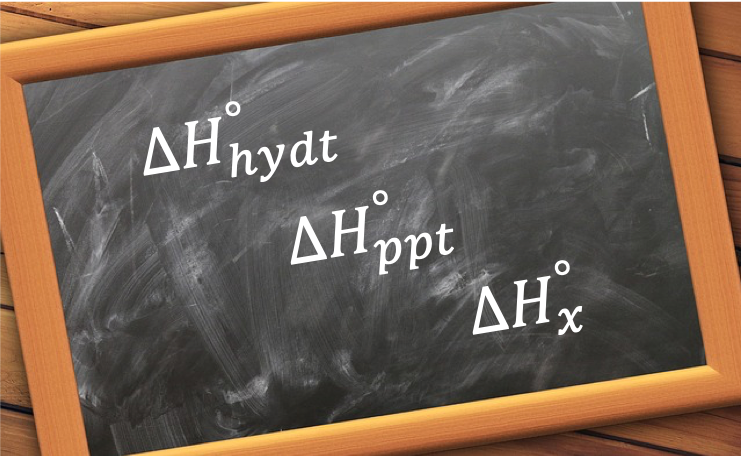The standard enthalpy change of vaporisation, ΔHvap o, is the change in enthalpy when one mole of a substance in the gaseous state is formed from the same substance in its liquid state under standard conditions.

Vaporisation reactions are always endothermic, e.g.
Even though ΔHvap o = 44.0 kJmol-1 is the standard enthalpy change of vaporisation of water at 298.15 K, many data sets of the standard enthalpy change of vaporisation of substances are quoted at the boiling points of those substances. In general, the standard enthalpy change of vaporisation of a substance is higher than the standard enthalpy change of fusion of that substance since molecules are separated further apart from one another from the liquid state to the gaseous state (more energy required) compared to the separation from the solid state to the liquid state. The standard enthalpy change of condensation of a substance, ΔHcon o, is the negative value of the standard enthalpy change of vaporisation of that substance.